1.What is the principle of morpholine as a metal corrosion inhibitor and what are its advantages?
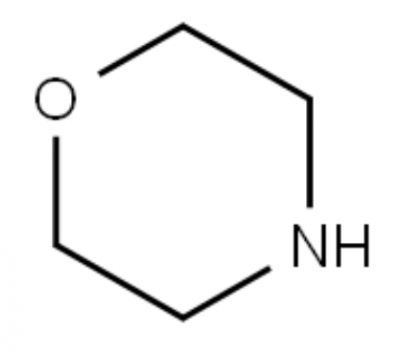
The reason why morpholine can be used as a metal corrosion inhibitor is that 1. the nitrogen and oxygen atoms in the morpholine molecule have lone pairs of electrons, which can form coordination bonds with the empty d orbitals of the metal surface (such as iron and copper), thereby adsorbing on the metal surface to form a dense monomolecular protective film. This film can effectively block contact with corrosive media such as water, oxygen, Cl⁻, and slow down electrochemical corrosion reactions (such as anodic oxidation and cathodic reduction). 2. Morpholine is weakly alkaline in water (pKa≈8.4), which can neutralize acidic corrosive environments and inhibit hydrogen ions (H⁺). It can corrode metals, especially in boilers or circulating water systems, by maintaining neutral or weakly alkaline conditions and slowing down the dissolution of metals. 3. Morpholine has a certain volatility and can form a gas-phase protective layer in closed systems (such as steam pipes) to inhibit the corrosion of metals by condensed water.
Morpholine is an environmentally friendly corrosion inhibitor, especially suitable for long-term protection of equipment in petrochemical, power, shipbuilding and other industries. The trend of environmentally friendly corrosion inhibitors replacing traditional products is obvious, and the market potential is great.
2.What role does N-methylmorpholine play in the production of polyurethane (PU) foam and what is the principle?
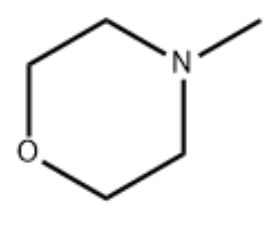
N-Methylmorpholine (NMM) mainly acts as a tertiary amine catalyst in the production of polyurethane (PU) foam. Its core function is to regulate the balance between foaming reaction and gelling reaction, thereby affecting the structure and performance of the foam. Specifically, there are about three functions: 1. Catalyze the foaming reaction, promote the reaction of isocyanate (such as MDI or TDI) with water, and generate carbon dioxide (CO₂) and polyurea structure. CO₂ acts as a foaming gas to form the porous structure of the foam. 2. Catalyze the gelling reaction, accelerate the polycondensation reaction of isocyanate and polyol, form the urethane main chain of polyurethane, and enhance the mechanical strength and network structure of the foam. 3. Balance the reaction rate, and control the pore structure (open or closed), density and elasticity of the foam by regulating the rates of foaming (gas generation) and gelling (network curing).
The principle is: 1. Alkaline catalytic mechanism, the nitrogen atom of N-methylmorpholine carries a lone pair of electrons, which acts as a proton acceptor and combines with the acidic hydrogen of isocyanate (such as the NCO group) to form a transition state intermediate, reducing the activation energy of the reaction, thereby accelerating the reaction. 2. Foaming reaction (water and isocyanate): NMM preferentially catalyzes the reaction to generate CO₂, promoting bubble nucleation and expansion, gel reaction (polyol and isocyanate): It also catalyzes the formation of carbamate to ensure that the foam quickly solidifies and sets after expansion. 3. NMM has moderate volatility and can escape the system in the later stage of the reaction, reducing the impact of residues on foam properties.
3.Why can N-methylmorpholine-N-oxide be used as the preferred solvent in the production of Lyocell fiber? What are the reasons?
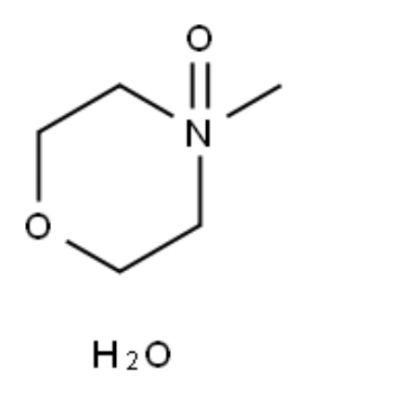
N-methylmorpholine-N-oxide (NMMO) is the preferred solvent in Lyocell fiber production mainly based on the following key reasons:
1. Efficient cellulose dissolving ability, breaking the hydrogen bond network: There are a large number of hydrogen bonds between cellulose molecules, which makes it difficult to dissolve in ordinary solvents. The strong polarity of NMMO and the oxygen atoms in the molecule can combine with cellulose hydroxyl groups through hydrogen bonds, effectively destroying the crystalline structure of cellulose and dissolving it. At high temperatures (about 80-120°C), NMMO can form a stable cellulose solution (concentration can reach 10-15%), meeting the requirements of the spinning process.
2. Environmental friendliness, NMMO is non-toxic or low-toxic. Compared with the traditional viscose method (using toxic substances such as CS₂), it greatly reduces environmental pollution. The recovery rate of NMMO is as high as more than 99%, and the production wastewater is very small, which meets the requirements of sustainable manufacturing.
3. Process compatibility, mild processing conditions: NMMO-cellulose solution can be directly dry-sprayed and wet-spun in a molten state without complex chemical derivatization (such as the yellowing reaction of the viscose method), the process is shorter, and the solution is stable: NMMO is not easy to decompose at high temperatures, the solution viscosity is moderate, which is conducive to spinning and forming, and the fiber structure is uniform.
3. Economic feasibility, controllable solvent cost: Although the unit price of NMMO is high, its high recovery rate reduces the long-term production cost, and the equipment is less corrosive: Compared with some ionic liquids, NMMO is less corrosive to equipment and extends the life of the equipment.
4. Optimized fiber performance, the NMMO dissolution process has little damage to the degree of polymerization of cellulose, and the resulting Lyocell fiber has high strength and good hygroscopicity, and its performance is close to that of natural cotton fiber, which is better than traditional regenerated cellulose fiber.
4.What is the relationship between morpholine, N-methylmorpholine, and N-methylmorpholine-N-oxide, and what are the differences in their uses?
Morpholine, N-methylmorpholine and N-methylmorpholine-N-oxide are three closely related organic compounds, which are gradually derived in structure and have different uses.
Morpholine (Morpholine), a six-membered heterocyclic compound, contains one oxygen atom and one nitrogen atom, and has a structural formula of C₄H₉NO. The nitrogen atom is unsubstituted (NH group), N-methylmorpholine (N-Methylmorpholine, NMM), a methyl substituent (N-CH₃) is introduced on the nitrogen atom of morpholine, with a structural formula of C₅H₁₁NO, and its alkalinity is slightly enhanced compared with morpholine, N-methylmorpholine-N-oxide (N-Methylmorpholine-N-oxide, NMO), the nitrogen atom of N-methylmorpholine is further oxidized to N-oxide (N⁺-O⁻ group), with a structural formula of C₅H₁₁NO₂, and its polarity is significantly improved.
Morpholine is more suitable for 1. As an industrial corrosion inhibitor for boiler water treatment systems and circulating water systems of steam power plants. 2. As a polymer synthesis catalyst for polyamide resin (nylon) polycondensation reaction and polyurethane foaming. 3. As a rubber vulcanization accelerator.
N-methylmorpholine is more suitable for 1. As a non-nucleophilic alkaline solvent. 2. Polar aprotic solvents for the polymerization of polyurethane (PU) or polyimide (PI). 3. Industrial cleaning agents (such as metal degreasing agents), emulsion polymerization aids.
N-methylmorpholine-N-oxide is more suitable for 1. Oxidation reaction co-oxidant. 2. Lyocell fiber (environmentally friendly artificial fiber) production. 3. Oxidation of specific functional groups in drug synthesis (such as alcohol → ketone, thioether → sulfoxide).
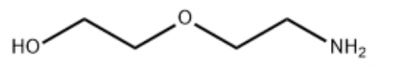
5.What is the principle of using diglycolamine for acid gas treatment and what are its advantages?
Principle: The amino group (-NH₂) in the DGA molecule reacts with acidic gases to generate reversible salts (such as carbamate or hydrogen sulfide), absorbs acidic gases at low temperature/high pressure, and then releases the gas by heating (regeneration), and DGA regains activity and is recycled.
Advantages:
1. High absorption capacity: DGA has a significantly higher absorption capacity for acidic gases than traditional amines (such as MEA and DEA), especially under high acidic gas partial pressure, which can reduce the amount of solvent circulation, equipment size and energy consumption.
2. Low regeneration energy consumption: Due to the low binding energy between DGA and acidic gases, the regeneration temperature (about 110–120°C) is lower than that of MEA (about 120–140°C), which saves steam consumption and reduces operating costs.
3. Anti-degradation and low corrosion: DGA is not easily oxidized or thermally degraded, which prolongs the life of the solvent, reduces the generation of byproducts (such as thermally stable salts), is less corrosive to equipment than MEA, and can reduce the amount of corrosion inhibitors and maintenance costs.
4. Strong adaptability, maintaining good fluidity at low temperatures (such as cold environments), suitable for harsh working conditions, has a certain ability to remove organic sulfur (such as mercaptans, COS), and is suitable for natural gas or refinery gas containing a variety of impurities.
5. Environmental protection and economy, low solvent loss, low volatility (low vapor pressure), reduced environmental pollution, low comprehensive energy consumption, and long-term operating costs are better than traditional amine methods.